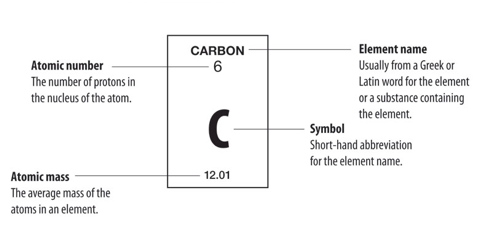 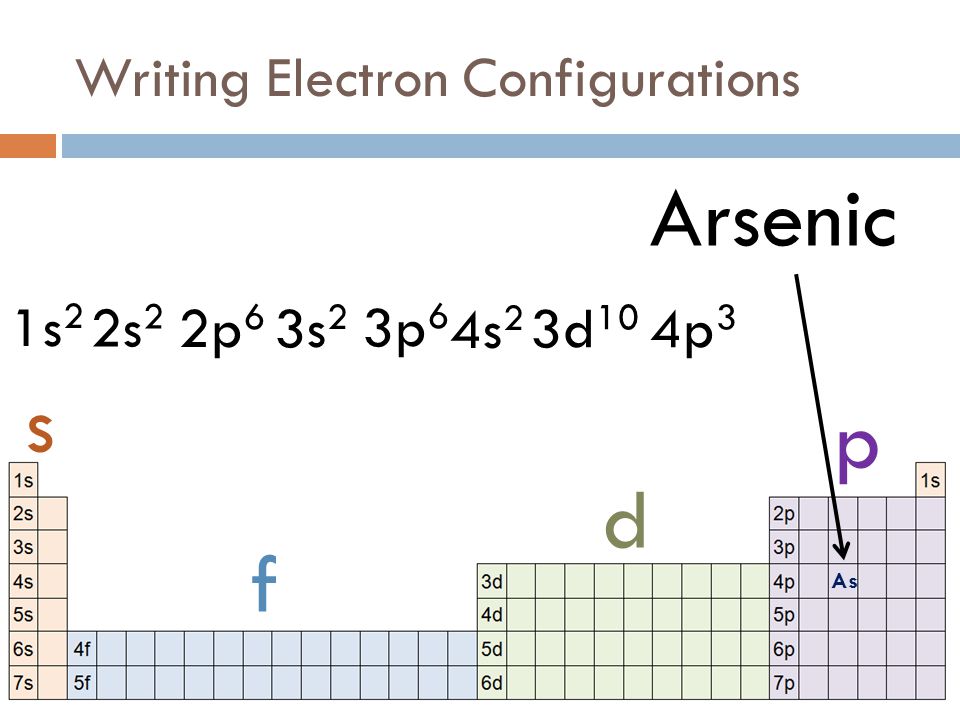
It doesn't matter which period the element's in, if it's in one of the main groups, so we're not counting the transition metals or the inner transition metals, we can look at the group number to determine how many valance electrons are present. And all electrons in partially filled sub-levels, so, for example, a sodium atom, which we just looked at, has one valence electron and that valence electron is in the 3s orbital. For the main group elements, the valence electrons are the highest energy s electrons. Valence electrons are very easy to count for the main group elements. That's the electron that leaves the sodium when the neutral sodium becomes sodium cation. Sodium has 1 valence electron, which is in a 3s orbital. There are 10 electrons in neon, and those are all core electrons of sodium. So, for example, if I had a sodium atom, I can write the electron configuration using the noble gas core of radiation, and the core electrons are summarized with just this symbol for neon in brackets. And it's the outermost valence electrons that leave the atom if it becomes a cation. It's those outermost valence electrons that are involved in chemical bonding. Those are the ones I've been considering when we talked about properties like ionization energy. The valence electrons are the outer most electrons. The core electrons are those bound inner electrons that are at the lower energy level, that are not involved in chemical reactions. Electrons can be divided into two types, the core electrons, and valence electrons. 
We did that, for example, when we were calculating formal charge in some of the molecules that I've drawn out. I know that, throughout the course, I've already mentioned, sometimes, the word, valence electron. The last thing I'd like to talk about is different types of electrons.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
